Structural Basis of the γ-Lactone-Ring Formation in Ascorbic Acid Biosynthesis by the Senescence Marker Protein-30/Gluconolactonase | PLOS ONE

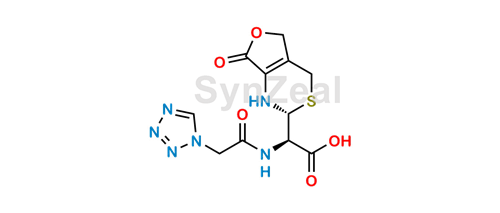
New structural scaffold 14-membered macrocyclic lactone ring for selective inhibitors of cell wall peptidoglycan biosynthesis in Staphylococcus aureus | The Journal of Antibiotics

Mild and Chemoselective Lactone Ring-Opening with (TMS)ONa. Mechanistic Studies and Application to Sweroside Derivatives | The Journal of Organic Chemistry
Structural Basis of the γ-Lactone-Ring Formation in Ascorbic Acid Biosynthesis by the Senescence Marker Protein-30/Gluconolactonase | PLOS ONE
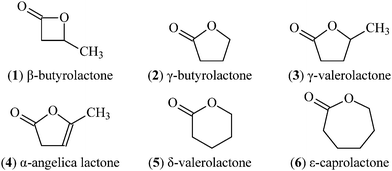
Degradable polymers from ring-opening polymerization of α-angelica lactone , a five-membered unsaturated lactone - Polymer Chemistry (RSC Publishing) DOI:10.1039/C1PY00067E

Controlled Random Terpolymerization of β-Propiolactone, Epoxides, and CO2 via Regioselective Lactone Ring Opening | CCS Chem
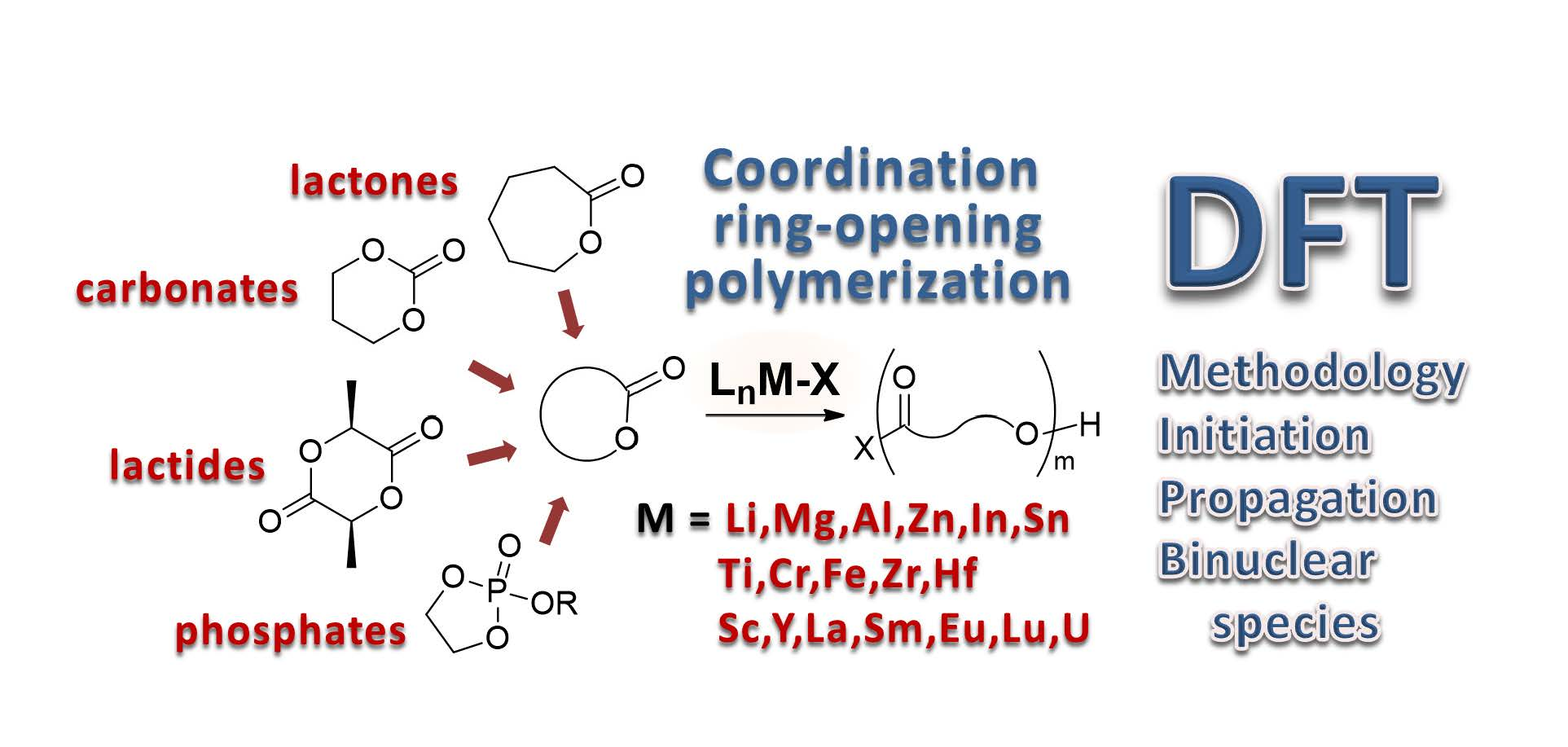
Molecules | Free Full-Text | Coordination Ring-Opening Polymerization of Cyclic Esters: A Critical Overview of DFT Modeling and Visualization of the Reaction Mechanisms

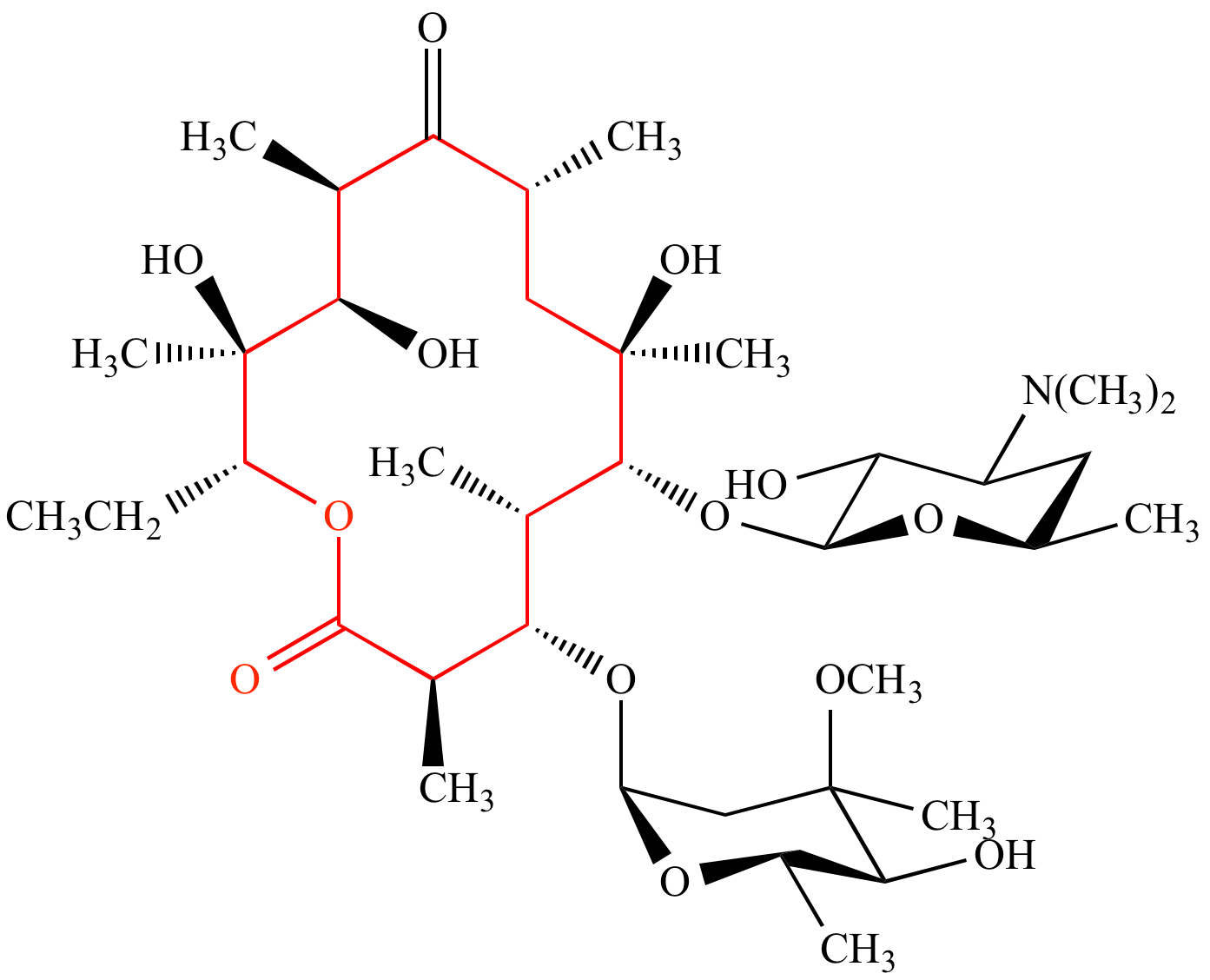
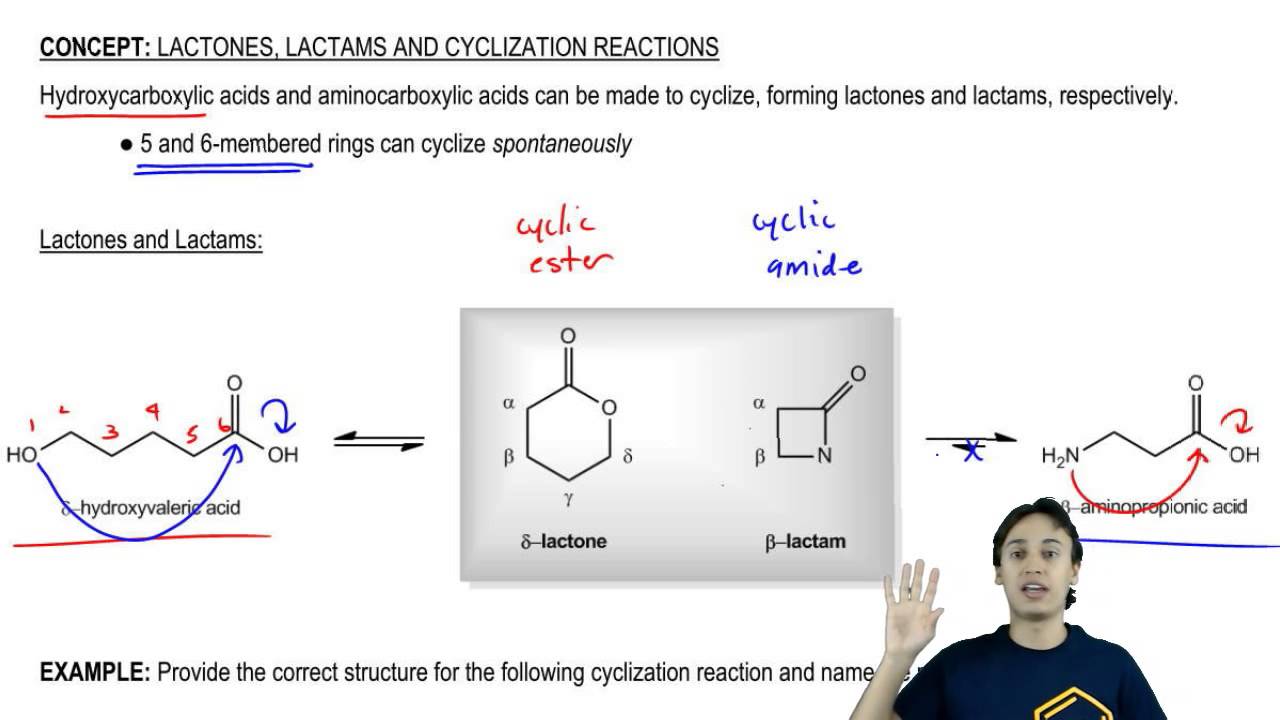
Lactones: Classification, synthesis, biological activities, and industrial applications - ScienceDirect

Organocatalysis applied to the ring‐opening polymerization of β‐lactones: A brief overview - Khalil - 2019 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library
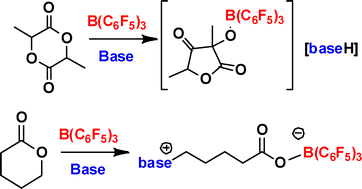
Ring openings of lactone and ring contractions of lactide by frustrated Lewis pairs - Dalton Transactions (RSC Publishing)

Why does ring opening reaction (of lactones) often follow nucleophilic pathway and not electrophilic? | ResearchGate

Synthesis of New Camptothecin Analogues with the E‐Lactone Ring Replaced by α,β‐Cyclohexenone - Bacherikov - 2006 - European Journal of Organic Chemistry - Wiley Online Library

Mild and Chemoselective Lactone Ring-Opening with (TMS)ONa. Mechanistic Studies and Application to Sweroside Derivatives | The Journal of Organic Chemistry